1s2 2s2 2p3 Oxygen configuration. 1s2 2s2 2p3 potassium.

Electron Configuration Ppt Download
The electrons from the electron configuration that are part of the Lewis structure of N are 2s2 2p3.
. 2 2 6 10 electrons total. Atoms of which element has the following electron configuration. In order to maximize energy these 14 electrons can be accommodated in the different molecular orbitals.
The electron configuration shows that arrangement of electrons in an atomElectrons in atoms are arranged in orbitalsThe electron configuration of Nitrogen is 1s2 2s2 2p3. Tap card to see definition. Experts are tested by Chegg as specialists in their subject area.
So an antimony atom with charge 2 has an electron configuration of 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p1. How many more electrons does nitrogen need to satisfy the octet rule. The first two electrons will go in the 1s orbital.
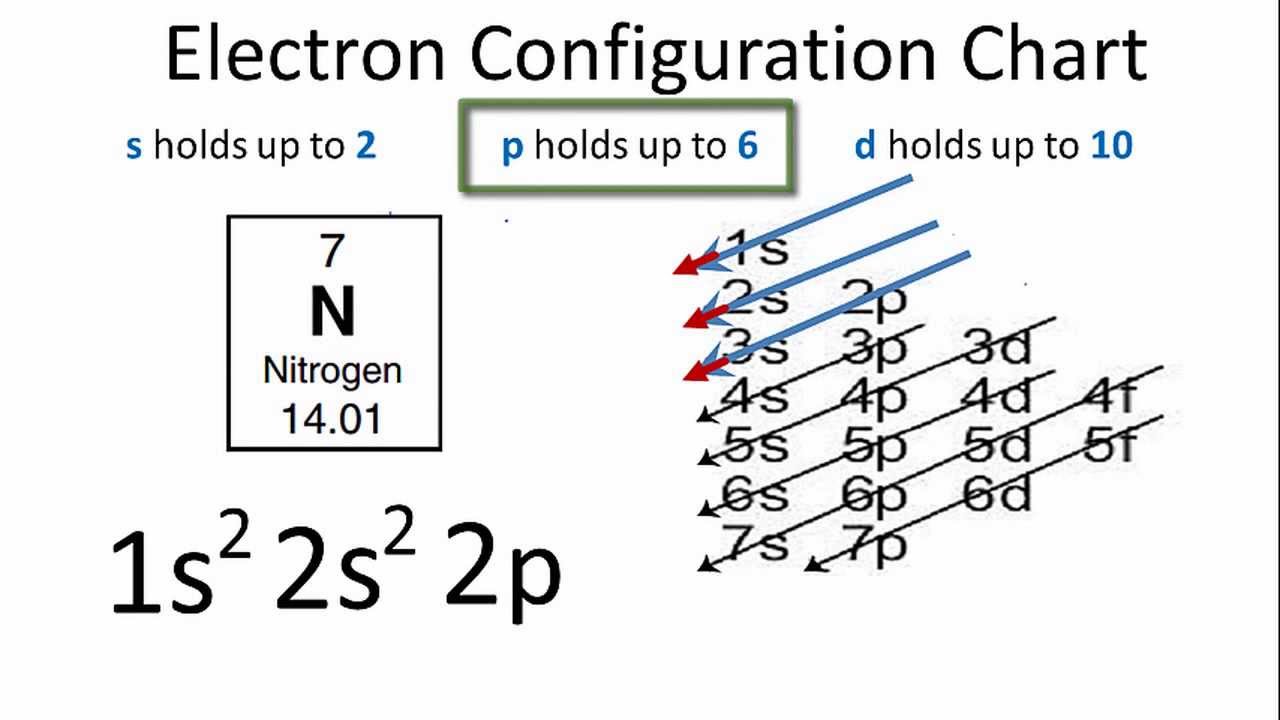
The electron configuration of nitrogen atomic number 7 is 1s2 2s2 2p3More precisely it is 1s2 2s2 2px12py1 2pz1. The atomic number of Nitrogen is 7 electronic configuration is 1s22s22p3. What electron configuration is 1s22s22p63s23p3.
1s2 2s2 2p6 3s3 3p6 4s2 3d7. The electronic configuration of nitrogen is 1s2 2s2 2p3. We review their content and use your feedback to keep the quality high.
For example here is a simple electron configuration. Element with electron configuration 1s2 2s2 2p3 is Nitrogen N It has two electrons in 1s two in 2s and three in 2p arbitrarily two in 2px and 1 in 2py 2. What element has an electron configuration of 1s2 2s2 2p6 3s2 3p6 3d10 4s2.
The total number of electrons present in the nitrogen molecule N2 is 14. How many of nitrogens electrons have ℓ 1. This configuration shows that there are 2 electrons in the 1s orbital set 2 electrons in the 2s orbital set and 6 electrons in the 2p orbital set.
Those are the small number placed at the top after the letters. Click card to see definition. The electronic configuration of an element is the arrangement of its electrons in its atomic orbitals.
1 2 Based on this what can you infer about the reactivity of helium and neon. Given that the electron configuration for phosphorus is 1s22s22p63s23p3 answer the following. The electron configuration of nitrogen is 1s2 2s2 2p3.
The closest noble gas neon Ne has the electronic configuration 1s2 2s2 2p6. Which one is correct electron configuration for a neutron atom of Nitrogen in excited state. To achieve a stable gas configuration nitrogen needs to have a fulfilled p orbital.
Nitrogen has a 1s 2 2s 2 2p 3 electron configuration. The electrons that appear in the Lewis structure are the outermost electrons in the. 1s2 2s2 2p5 Neon configuration.
The electron configuration of nitrogen is 1s2 2s2 2p3 How many more electrons does nitrogen need to satisfy the octet. 1s2 2s2 2p4 Fluorine configuration. To figure this out the element with the electron config of we first count the electrons.
Since 1s can only hold two electrons the next 2 electrons for N goes in the 2s orbital. The final electron configuration is a single string of orbital names and superscripts. 1s2 2s2 2p6 3s2 2p3.
Identify the inner electrons outer electrons and valence electrons in each electron configurations. Nitrogen molecule N2 The electronic configuration of nitrogen Z7 1s2 2s2 2px12py12pz1. Thus the p orbital is the outermost shell.
Who are the experts. Thus nitrogen has a half-filled P orbital which is comparatively more stable. How does oxygen obey the octet rule when reacting to form compounds.
The electron configuration for Copper Co is. Using the blocks in the periodic table we can write the electron configuration of bromine as. It gives up electrons.
1s2 2s2 2p5 Your answer is given below.

Nitrogen Electron Configuration Youtube
What Is The Electronic Configuration Of Nitrogen Using The Electron In Box Notation Quora

Which Element Has The Electron Configuration Of 1s2 2s2 2p3 Youtube
0 Comments